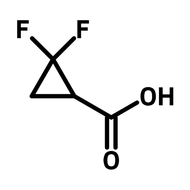
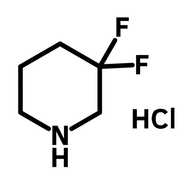
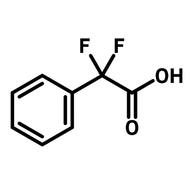
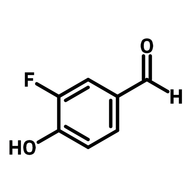
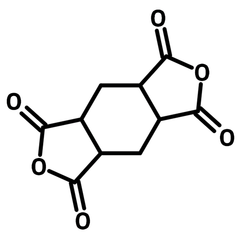
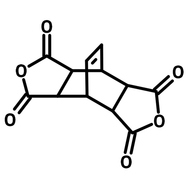
1,2,4,5-cyclohexanetetracarboxylic dianhydride (HPMDA)
CAS Number 2754-41-8
Chemistry Building Blocks, COF Ligands, Diamines and Dianhydrides, Heterocyclic Building Blocks, Materials, Monomers, Porous Organic Frameworks
An alicyclic dianhydride building block
For polyimides and polyesters synthesis to be applied as photochromic materials, proton conducting membranes, and bioimaging polymers
1,2,4,5-Cyclohexanetetracarboxylic dianhydride (HPMDA, CAS number 2754-41-8) has a cyclohexane centre with two carboxylic anhydrides substituents sitting opposite to each other. HPMDA reacts with amines for polyimides or alternatively with alcohols for polyesters. The non-aromatic cyclohexane centre provides local excited state to the synthesized photochromic polyimides showing large contrast of the photo-switching effect (compared to fully aromatized polyimides). The polyimides derived from 1,2,4,5-cyclohexanetetracarboxylic dianhydride exhibit high proton conductivity (0.2 ± 0.01 S/cm at 298 K and 95% related humidity). Like other polyimides, the polyimides have excellent chemical and thermal resistance and are used in optical fibre claddings and substrates for microelectronics.
HPMDA can be used to synthesize polyesters that have four branches (dianhydride is hydrolysed to four carboxylic acids) for multiple functionalities, such as biodegradability, bioimaging and photoactivity.
General Information
| CAS Number | 2754-41-8 |
| Chemical Formula | C10H8O6 |
| Full Name | 1,2,4,5-Cyclohexanetetracarboxylic dianhydride |
| Molecular Weight | 224.17 g/mol |
| Synonyms | Hexahydro-1H,3H-furo[3,4-f][2]benzofuran-1,3,5,7-tetrone, Tetrahydrobenzo[1,2-c:4,5-c']difuran-1,3,5,7(3aH,7aH)-tetraone |
| Classification / Family | Dianhydride building block, Ionic conducting membranes, Polyimides, Polyesters |
Chemical Structure

Product Details
| Purity | >99% |
| Melting Point | Tm = 240 °C |
| Appearance | White to off-white powder/crystal |
MSDS Documentation
 1,2,4,5-cyclohexanetetracarboxylic dianhydride (HPMDA) MSDS Sheet
1,2,4,5-cyclohexanetetracarboxylic dianhydride (HPMDA) MSDS Sheet
Literature and Reviews
-
Functional polyimides based on diamine containing diarylethylene moieties and their photochromic mechanism studies, W. Huang et al., Polym. Chem., 11, 6701(2020); DOI: 10.1039/d0py01084g.
-
Lyotropic liquid crystalline property and organized structure in high proton-conductive sulfonated semialicyclic oligoimide thin film, Y. Yao et al., ACS Omega, 8, 7470-7478(2023); DOI: 10.1021/acsomega.2c06398.
-
Optically transparent sulfur-containing polyimide-TiO2 nanocomposite films with high refractive index and negative pattern formation from poly(amic acid)-TiO2 nanocomposite film, J.-G. Liu et al., Chem. Mater., 20, 273-281(2008); DOI: 10.1021/cm071430s.
-
Progress in aromatic polyimide films for electronic applications: preparation, structure and properties, Z. Wu et al., Polymers, 14, 1269(2022); DOI: 10.3390/polym14061269.
-
Quartz fiber cloth-reinforced semi-aromatic thermosetting polyimide composite with high-frequency low dielectric constant, Z.-h. Wang et al., High Perform. Polym., 32(1), 91-102(2020); DOI: 10.1177/0954008319853028.
- Self-sacrificially degradable pseudo-semiconducting polymer nanoparticles that integrate NIR-II fluorescence bioimaging, photodynamic immunotherapy, and photo-activated chemotherapy, D. Tang et al., Adv. Mater., 34(34), 2203820(2022); DOI: 10.1002/adma.202203820.
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
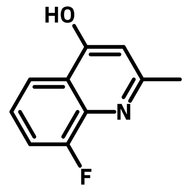
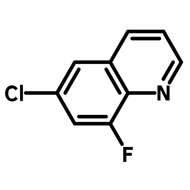
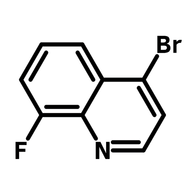
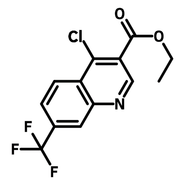
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)