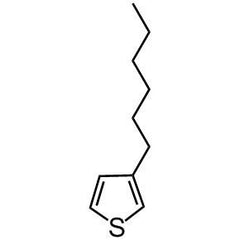
3-Hexylthiophene
CAS Number 1693-86-3
Chemistry Building Blocks, Heterocyclic Building Blocks, Materials, Monomers
3-Hexylthiophene, for the synthesis of P3HT
High-quality and high-purity monomer available online
3-Hexylthiophene, CAS number 1693-86-3, is the intermediate for the synthesis of poly(3-hexylthiophene), referred as P3HT. To date, it is the most studied polymer for polymer solar cells. The efficiency of a P3HT/PCBM solar cell is typically 4-5 %, but with new fullerene materials developed to closely match the energy levels of P3HT (HOMO 5.0 eV, LUMO 3.0 eV), device performances have been pushed to 6.5% [1].
The synthesis of P3HT is relatively easy and short, only a 3-4 step synthesis is required [2, 3, 4]. The challenge of P3HT is that it only absorbs a narrow band of solar spectrum, so it has little room for improvement in terms of performance efficiency.
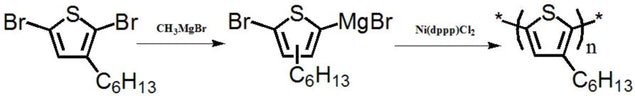
A synthesis precusor
For poly(3-hexylthiophene) (P3HT)
Hexylthiophene building block
For semiconductors, OFETs, and solar cells
Worldwide shipping
Quick and reliable shipping
High purity
>99% High purity
General Information
| CAS Number |
1693-86-3 |
| Chemical Formula | C10H16S |
| Molecular Weight | 168.30 g/mol |
| Synonyms | 1-(Thien-3-yl)hexane 3-n-Hexylthiophene |
| Classification / Family | Monomers, Building blocks, Thiophene, Heterocycles, Chemical synthesis for low band gap polymers, Intermediates for OFETs, OLED, Organic Photovoltaics, Polymer solar cells |
Chemical Structure
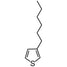
Product Details
| Purity | 99% |
| Boiling Point | 65 °C at 0.45 mmHg (lit.) 299 °C at 760 mmHg (1 atm, lit.) |
| Density | 0.936 g/cm3 |
| Appearance | Colourless/pale yellow liquid |
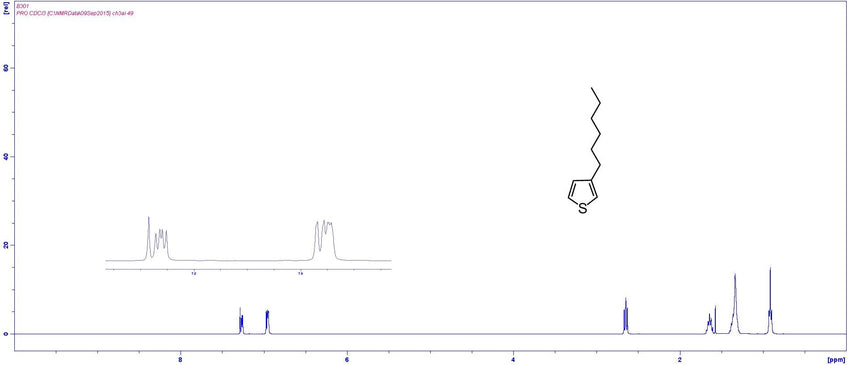
NMR Characterisation
MSDS Documentation
Literature and Reviews
- 6.5% Efficiency of Polymer Solar Cells Based on poly(3‐hexylthiophene) and Indene‐C60 Bisadduct by Device Optimization, G. Zhao et al., Adv. Mater., 22, 4355–4358 (2010).
- Regiocontrolled Synthesis of Poly(3-alkylthiophenes) Mediated by Rieke Zinc: Their Characterization and Solid-State Properties, Chen et. al., J. Am. Chem. Soc., 117 (1), pp 233–244 (1995).
- A Simple Method to Prepare Head-to-Tail Coupled, Regioregular Poly(3-alkylthiophenes) Using Grignard Metathesis., R. S. Loewe et al., Adv. Mater., 11: 250–253 (1999)
- Synthesis and characterisation of telechelic regioregular head-to-tail poly(3-alkylthiophenes), A. Iraqi et. al., J. Mater. Chem., 8, 25-29 (1998).
- Charge Transport and Photocurrent Generation in Poly(3-hexylthiophene):Methanofullerene Bulk-Heterojunction Solar Cells, D. Valentin et al, Adv. Funct. Mater., 16, 699–708 (2006).
- Soluble and processable regioregular poly(3‐hexylthiophene) for thin film field‐effect transistor applications with high mobility, Z. Bao et al., Appl. Phys. Lett. 69, 4108 (1996).
- Dependence of Regioregular Poly(3-hexylthiophene) Film Morphology and Field-Effect Mobility on Molecular Weight, R. J. Kline, Macromolecules, 38 (8), pp 3312–3319 (2005).
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
 3-Hexylthiophene MSDS sheet
3-Hexylthiophene MSDS sheet
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
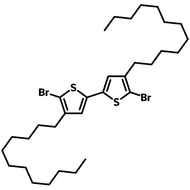
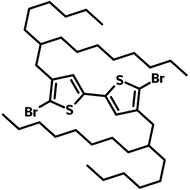
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
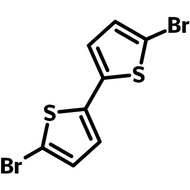
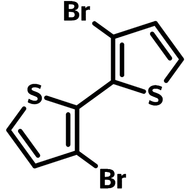
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)