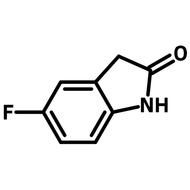
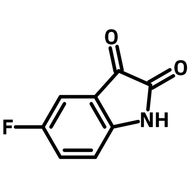
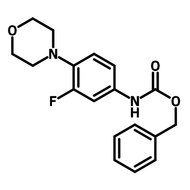
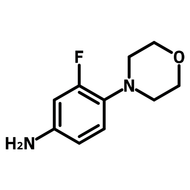
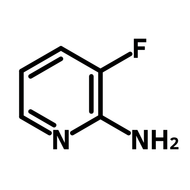
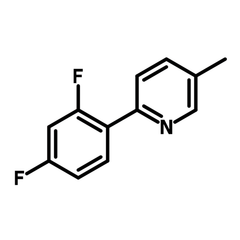
2-(2,4-Difluorophenyl)-5-methylpyridine
CAS Number 583052-21-5
Chemistry Building Blocks, Fluorinated Building Blocks, Heterocyclic Building Blocks, Monomers
A difluorinated heterocyclic building block
As cyclometalating ligand to form coordinative complexes in application of OLEDs, DSSCs and photocatalysis reactions
2-(2,4-Difluorophenyl)-5-methylpyridine (DFPMPy), CAS number 583052-21-5, is a difluorinated heterocyclic derivative of arylpyridine with enhanced solubility by the methyl group. DFPMPy is often used as a cyclometalating ligand with the nitrogen electron lone pair and the delocalized π-electron cloud in the benzene ring being coordinated to a metal centre. The metal-ligand complexes have demonstrated excellent photocatalytic capability in visible-light photoredox reactions. During the photoredox process, an electron migrates from the metal centre to the ligands, also known as metal-ligand charge transfer (MLCT) after irradiation as the complex reaches the excited state. After the single-electron transfer, the excited complex regains an electron from another reagent and restart the photoredox catalytic cycle.
These complexes are also synthesized for light-emitting electrochemical cells with photoluminescent quantum yield up to 93%. The fluorine-substituents lower the highest occupied molecular orbital (HOMO) energy level resulting shifts in the emission spectrum. Arylpyridine-iridium complexes are widely investigated in water splitting to generate hydrogen. As photosensitizers, these complexes are also used in dye-sensitized solar cells (DSSCs).
Multiple functional groups
For facile synthesis
Fluorinated phenylpyridine building block
For drug discoveries, solar cells, and photocatalists
Worldwide shipping
Quick and reliable shipping
High purity
>97% High purity
General Information
| CAS Number | 583052-21-5 |
| Chemical Formula | C12H9F2N |
| Full Name | 2-(2,4-Difluorophenyl)-5-methylpyridine |
| Molecular Weight | 205.21 g/mol |
| Synonyms | DFPMPy |
| Classification / Family | Pyridine derivatives, Fluorinated building blocks, Heterocyclic building block, Dyes, DSSCs, Photocatalyst ligands, OLEDs |
Chemical Structure

Product Details
| Purity | >97% |
| Melting Point | Tm = 55 °C |
| Appearance | White/off-white to yellow to light brown powder/crystal |
MSDS Documentation
 2-(2,4-Difluorophenyl)-5-methylpyridine MSDS Sheet
2-(2,4-Difluorophenyl)-5-methylpyridine MSDS Sheet
Literature and Reviews
-
Visible light driven generation and alkyne insertion reactions of stable bis-cyclometalated Pt(IV) hydrides, D. Poveda et al., Chem. Sci., 11, 12095–12102(2020); DOI: 10.1039/d0sc04879h.
-
Cationic iridium (III) complexes bearing ancillary 2,5-dipyridyl(pyrazine) (2,5-dpp) and 2,2’:5’,2’’- terpyridine (2,5-tpy) ligands: synthesis, optoelectronic characterization and light-emitting electrochemical cells, K. Hasan et al.,
Dalton Trans., 43, 13672-13682(2014); DOI: 10.1039/C4DT02100B.
-
Generation of phosphoranyl radicals via photoredox catalysis enables voltage−independent activation of strong C−O bonds, E. Stache et al., ACS Catal., 8(12), 11134–11139(2018); DOI: 10.1021/acscatal.8b03592.
-
Intimate electronic coupling in cationic homodimeric iridium(III) complexes, A. Soliman et al., Dalton Trans., 41, 9382(2012); DOI: 10.1039/c2dt30836c.
-
Merging photoredox and nickel catalysis: decarboxylative cross-coupling of carboxylic acid with vinyl halides, A. Noble et al., J. Am. Chem. Soc., 137, 624-627(2015); DOI: 10.1021/ja511913h.
-
Recent advances in synthesis of molecular heteroleptic osmium and iridium phosphorescent emitters, M. Buil et al., Eur. J. Inorg. Chem., 2021(46), 4731–4761(2021); DOI: 10.1002/ejic.202100663.
-
Intermolecular crossed [2+2] cycloaddition promoted by visible-light triplet photosensitization: expedient access to polysubstituted 2-oxaspiro[3.3]heptanes, P. Murray et al., J. Am. Chem. Soc., 143(10), 4055–4063(2021); DOI:10.1021/jacs.1c01173.
-
The transformation and storage of solar energy: progress towards visible-light induced water splitting, E. Cline et al., Chimia, 63, 709-713(2009); DOI: 10.2533/chimia.2008.709.
- Tuning the emission of cationic iridium(III) complexes towards the red through methoxy substitution of the cyclometalating ligand, K. Hasan et al., Sci. Rep., 5, 12325(2015); DOI: 10.1038/srep12325.
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
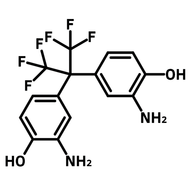
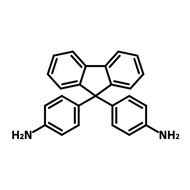
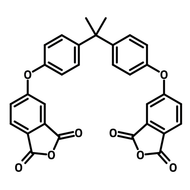
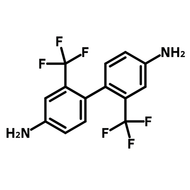
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)